Determine Density of Viscous Material
Written by Maynard R. Euverard former Consultant to the Paul N. Gardner Co., Inc.
Density (weight per unit volume) is one of the most important physical properties of coating materials and other liquids and semiliquids. Density is important because it is a first-line indication of the ratios of components that make up a material. Many materials are formulated and produced by weight and are sold by volume.
Accurately determining the density of nonviscous liquids is usually straightforward, involving weighing a standard volume of material. These nonviscous materials do not entrap air; the air rises and breaks free at the surface. However, if the material is viscous, the arrived-at density can be inaccurate. Viscous materials can entrap air, causing false volume readings.
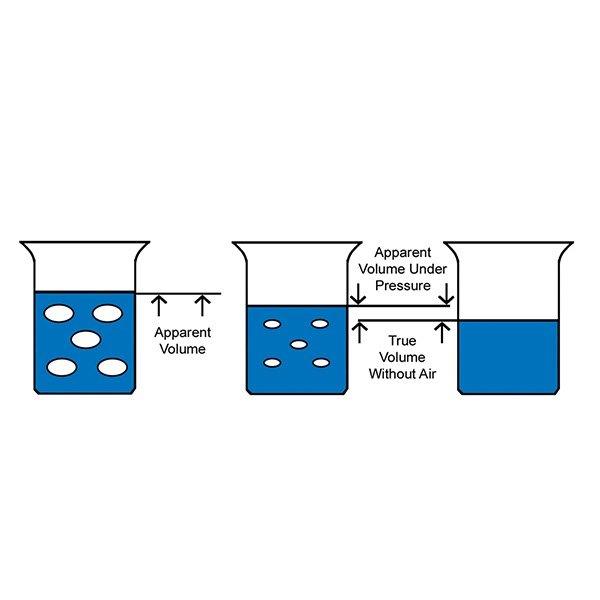
The false apparent volume of a viscous material containing entrapped air is shown in the image with the blue liquid. If pressure is exerted onto the material, the apparent volume will decrease due to compression of the entrapped air. The material's true volume would be without the entrapped air.
The density in pounds per gallon of nonviscous materials can be found readily using a standard "weight per gallon cup" and a scale capable of reading to the nearest tenth gram. The most widely accepted such cup accommodates a weight of distilled water at 77°F of 83.2 g. To convert the gram weight of the cup's contents to pounds per gallon, it is necessary only to divide by 10 (move the decimal one place to the left). This is because the absolute density of water at 77°F is 8.32 lb./gal.
Diluting and Mixing
The accurate volume determination of a viscous material with entrapped air can be determined by a method that involves diluting and mixing a sample of the material with an acceptable diluent (thinner).
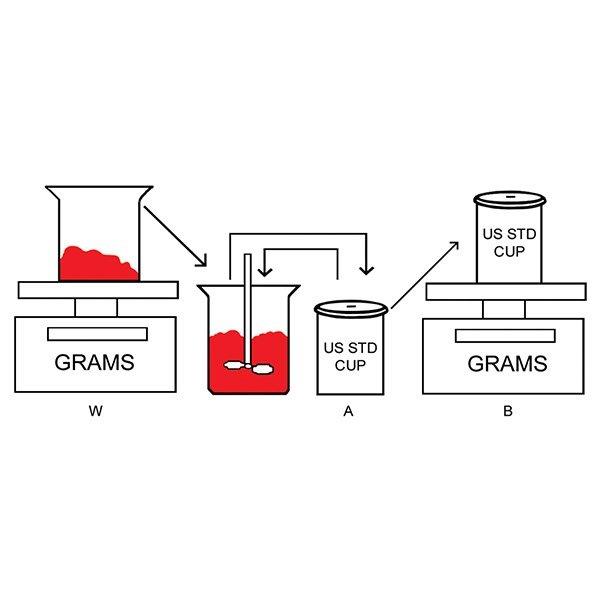
This diluting and mixing frees the viscous material sample of entrapped air. A standard weight-per-gallon cup can be filled with the diluted and mixed materials and weighed.The procedure to determine the density of a viscous material with entrapped air is illustrated in these drawings.
Mixing a diluent (A) with a sample of the viscous material (W) frees the entrapped air. The weight-per-gallon cup (B) contains a level-full amount of the diluent-viscous material mixture.
The true weight per gallon of the viscous material without entrapped air can then be calculated from the formula listed here.
X= (W x B) / (W + A - (10 x B))
| X | is the unknown lb/gal density of the viscous material. |
| W | is the measured gram weight of the viscous material sample. |
| A | is the gram weight of the diluent from a full weight-per-gallon cup. |
| 10 | is the multiplier to convert "B" into gram weight of a filled weight-per-gallon cup of the viscous material. |
Proof of the formula follows:
lb/gal = (grams x lb/gal) / (grams + grams - (K x lb/gal))
lb/gal = grams/10 This is the cup conversion factor.
Substituting the conversion factor in the original formula:
grams/10 = (grams x grams/10) / (grams + grams - K x grams/10)
K is the conversion factor equal to 10.
Substituting 10 for K:
grams/10 = (grams + grams/10) / (grams + grams - grams)
grams/10 = grams/10
1 = 1
The method has been confirmed in the laboratory by using two materials of widely different densities. The amount of the unknown material (W) was varied greatly. The calculated weight per gallon (X) throughout the range of 25 grams to 125 grams of the unknown was always within 0.5% of the known true value.
The weight-per-gallon cups are available in corrosion-resistant materials. One such cup is produced with a sidewall of series 316 stainless steel and a machined bottom and closely fitted lid of series 416 stainless steel. A radiused area where the bottom is secured to the sidewall provides ease of cleaning. The lid is formed with a small opening to permit discharge of excess material to ensure exact specified volume of the cup contents. The cups are warranted to be within 0.5% of specified volume. Cups are available that qualify for volume certification under MIL STD 45662 and exceed the requirements of ASTM methods.
Cups of other volumes are available, including a "mini" cup that is one-tenth the U.S. standard cup. Another cup readily converts pounds and imperial gallons. These cups can also be used in this indirect computational method of measurement provided the correct multiplier is used in the equation for "B."
Copied with permission from the March 1989 issue of Industrial Finishing Magazine Copyright© 1989, Hitchcock Publishing Co. All rights reserved.